Foram encontradas 80 questões.
Considere a sequência de reações orgânicas abaixo:
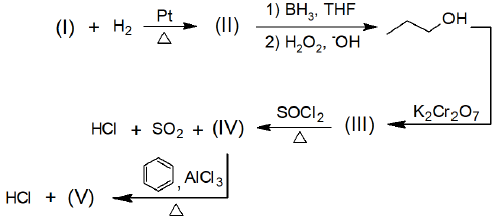
A opção que corresponde aos compostos de (I) a (V), respectivamente, é:
Provas
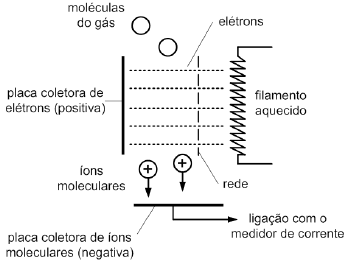
A figura abaixo esquematiza o funcionamento de um aparelho de ionização que pode ser útil para medir baixas pressões compreendidas entre 10–4 e 10–10 mmHg. Nesse dispositivo, elétrons partem de um filamento aquecido, atravessam uma rede cuja tensão fixa a energia do elétron, e atingem uma região do tubo sonda ligada ao sistema de alto vácuo cuja pressão se deseja medir. Esses elétrons ionizam espécies neutras presentes no tubo e formam íons positivos que são atraídos por uma placa coletora negativa. Além disso, produzem uma corrente que pode ser medida e correlacionada com a pressão do sistema de vácuo. Portanto, quanto mais baixa a pressão, menor o número de moléculas neutras e, consequentemente, menor o número de íons positivos formados no tubo.
O gráfico abaixo relaciona as primeiras energias de ionização aos números atômicos dos respectivos elementos.
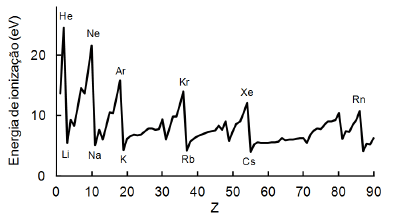
Um aparelho de ionização cuja energia eletrônica é 15 eV foi calibrado medindo-se a pressão de um sistema que continha vapor de sódio. Como a leitura do instrumento seria afetada se o vapor de sódio fosse substituído por neônio à mesma pressão?
Provas
Considere a representação da Tabela Periódica a seguir:
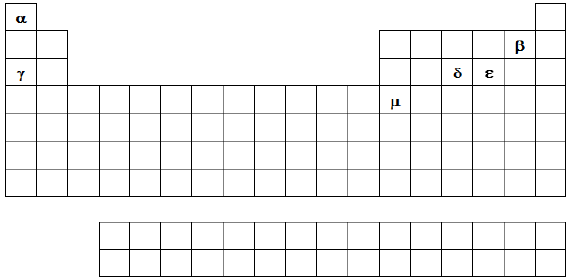
Com base nessa representação da tabela, avalie as asserções abaixo:
I. O composto de representação !$ δ \alpha_3 !$ é iônico;
II. A distribuição eletrônica [Ar] 4s2 3d8 pode representar o íon !$ \mu^{3+} !$;
III. O isótopo mais estável do elemento !$ γ !$ tem 12 nêutrons;
IV. Os elementos que apresentam, em seu estado fundamental, a distribuição eletrônica [Ne] 3s2 3py, com 1 !$ \le !$ y !$ \le !$ 6, são todos não metais;
V. O raio atômico de !$ ε !$ é menor que o raio atômico de !$ δ !$.
Assinale a alternativa que contém somente asserções corretas.
Provas
Assinale a alternativa correta.
Provas
A azitromicina é um potente antibiótico comercial. Sua estrutura molecular está mostrada abaixo:
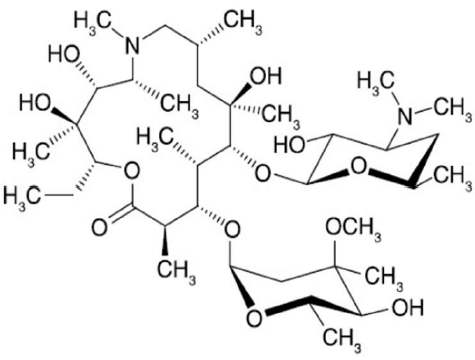
Considerando a estrutura acima, são feitas as seguintes afirmações:
I. Existem 2 átomos com hibridização sp2.
II. A molécula possui 18 carbonos quirais.
III. Éster, amina e éter são funções orgânicas encontradas na molécula.
Com base na análise das afirmações acima, assinale a opção correta:
Provas
Uma liga de cobre e prata, isenta de impurezas, é colocada em um recipiente contendo uma solução de ácido sulfúrico e água bromada, de modo que o gás que se desprende durante a reação é integralmente absorvido pela água bromada. Após a dissolução completa da liga, adiciona-se uma solução aquosa de BaCl2. Findo o procedimento, observa-se um precipitado que deve ser composto predominantemente por:
Provas
A respeito das reações abaixo:
I. !$ \overset{27}{13} !$!$ Al+ !$!$ \overset{4}{2} !$!$ \alpha \rightarrow !$!$ \overset{30}{15} !$!$ P+ !$!$ \overset{1}{0} !$!$ n !$
II. !$ \overset{1}{0} !$!$ n+ !$!$ \overset{235}{92} !$!$ U \rightarrow !$!$ \overset{142}{56} !$!$ Ba+ !$!$ \overset{x}{y} !$!$ Kr+ !$!$ 3 !$!$ \overset{1}{0} !$!$ n !$
III. !$ \overset{2}{1} !$!$ H+ !$!$ \overset{3}{1} !$!$ H \rightarrow !$!$ \overset{4}{2} !$!$ He + !$!$ \overset{1}{0} !$!$ n !$
Assinale a alternativa INCORRETA.
Provas
Considere que a superfície da Lua seja bombardeada a cada segundo por cerca de 100 bilhões de átomos de hidrogênio por cm2 em função da ação do “vento solar”. Supondo que esse fluxo se mantenha constante, a massa aproximada de hidrogênio, que atingirá 1 cm2 da Lua nos próximos 5 milhões de anos será:
(Dado: NA = 6,0.1023)
Provas
Texto 4
HOW MUCH CAN WE KNOW?
The reach of the scientific method is constrained by the limitations of our tools and the intrinsic impenetrability of some of nature's deepest questions.
“What we observe is not nature in itself but nature exposed to our method of questioning,” wrote German physicist Werner Heisenberg, who was the first to fathom the uncertainty inherent in quantum physics. To those who think of science as a direct path to the truth about the world, this quote must be surprising, perhaps even upsetting.
People will quickly counterstrike with something like: Why do airplanes fly or antibiotics work? Why are we able to build machines that process information with such amazing efficiency? Surely, such inventions and so many others are based on laws of nature that function independently of us. There is order in the universe, and science gradually uncovers this order.
No question about it: There is order in the universe, and much of science is about finding patterns of behavior—from quarks to mammals to galaxies—that we translate into general laws. We strip away unnecessary complications and focus on what is essential, the core properties of the system we are studying. We then build a descriptive narrative of how the system behaves, which, in the best cases, is also predictive.
Often overlooked in the excitement of research is that the methodology of science requires interaction with the system we are studying. We observe its behavior, measure its properties, and build mathematical or conceptual models to understand it better. We can see only so far into the nature of things, and our ever shifting scientific worldview reflects this fundamental limitation on how we perceive reality.
Just think of biology before and after the microscope or gene sequencing, or of astronomy before and after the telescope, or of particle physics before and after colliders or fast electronics. Now, as in the 17th century, the theories we build and the worldviews we construct change as our tools of exploration transform. This trend is the trademark of science.
Sometimes people take this statement about the limitation of scientific knowledge as being defeatist: “If we can’t get to the bottom of things, why bother?” This kind of response is misplaced. There is nothing defeatist in understanding the limitations of the scientific approach to knowledge. Science remains our best methodology to build consensus about the workings of nature. What should change is a sense of scientific triumphalism—the belief that no question is beyond the reach of scientific discourse.
[...]
Adaptado de GLEISER, Marcelo. How Much Can We Know? Nature, International Journal of Science. Disponível em: <https://www.nature.com/articles/d41586-018-05100-5>. Acesso em: 14/08/2019.
Choose the correct option.
Provas
Texto 4
HOW MUCH CAN WE KNOW?
The reach of the scientific method is constrained by the limitations of our tools and the intrinsic impenetrability of some of nature's deepest questions.
“What we observe is not nature in itself but nature exposed to our method of questioning,” wrote German physicist Werner Heisenberg, who was the first to fathom the uncertainty inherent in quantum physics. To those who think of science as a direct path to the truth about the world, this quote must be surprising, perhaps even upsetting.
People will quickly counterstrike with something like: Why do airplanes fly or antibiotics work? Why are we able to build machines that process information with such amazing efficiency? Surely, such inventions and so many others are based on laws of nature that function independently of us. There is order in the universe, and science gradually uncovers this order.
No question about it: There is order in the universe, and much of science is about finding patterns of behavior—from quarks to mammals to galaxies—that we translate into general laws. We strip away unnecessary complications and focus on what is essential, the core properties of the system we are studying. We then build a descriptive narrative of how the system behaves, which, in the best cases, is also predictive.
Often overlooked in the excitement of research is that the methodology of science requires interaction with the system we are studying. We observe its behavior, measure its properties, and build mathematical or conceptual models to understand it better. We can see only so far into the nature of things, and our ever shifting scientific worldview reflects this fundamental limitation on how we perceive reality.
Just think of biology before and after the microscope or gene sequencing, or of astronomy before and after the telescope, or of particle physics before and after colliders or fast electronics. Now, as in the 17th century, the theories we build and the worldviews we construct change as our tools of exploration transform. This trend is the trademark of science.
Sometimes people take this statement about the limitation of scientific knowledge as being defeatist: “If we can’t get to the bottom of things, why bother?” This kind of response is misplaced. There is nothing defeatist in understanding the limitations of the scientific approach to knowledge. Science remains our best methodology to build consensus about the workings of nature. What should change is a sense of scientific triumphalism—the belief that no question is beyond the reach of scientific discourse.
[...]
Adaptado de GLEISER, Marcelo. How Much Can We Know? Nature, International Journal of Science. Disponível em: <https://www.nature.com/articles/d41586-018-05100-5>. Acesso em: 14/08/2019.
According to the following passage from the text: “Just think of biology before and after the microscope or gene sequencing, or of astronomy before and after the telescope, or of particle physics before and after colliders or fast electronics.”, choose the correct option.
Provas
Caderno Container